Staff Writer Arifin Chowdhury, ’22
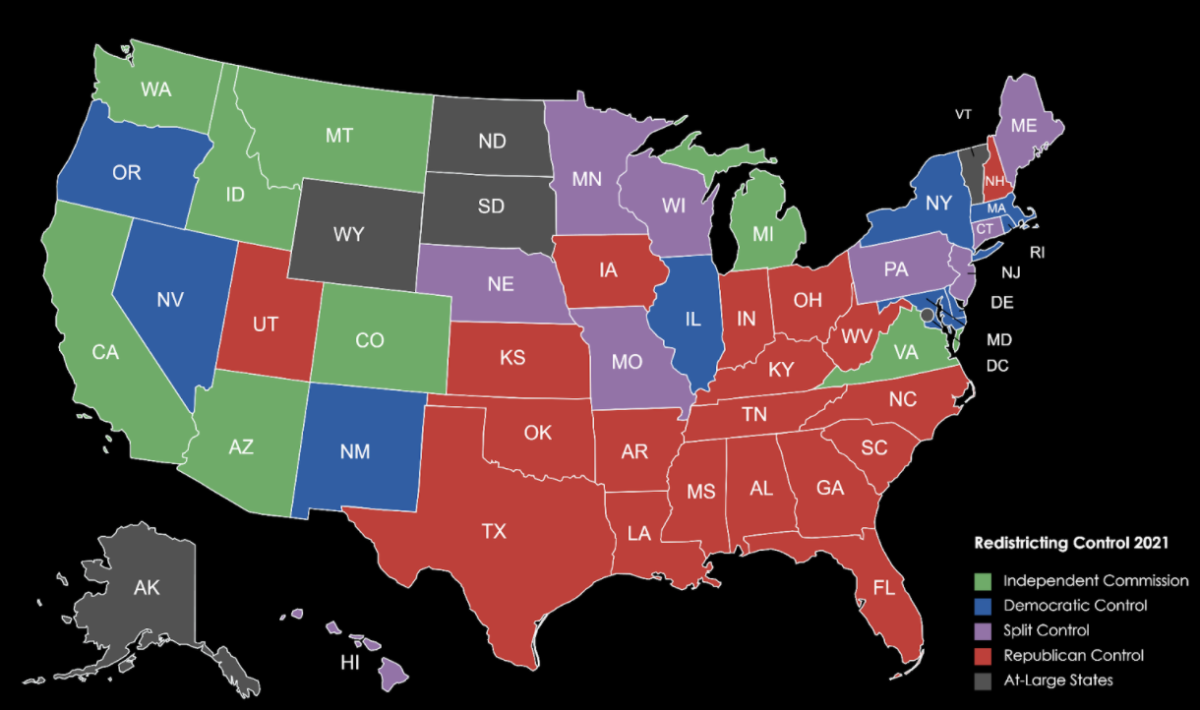
On December 18, the FDA approved Moderna’s vaccine for emergency use in the US. Initial allocations for the Pfizer and BioNTech vaccine provided enough doses for 170,000 New Yorkers. The vaccine is first being distributed to high-risk hospital workers, nursing home residents, nursing home staff, followed by all long-term and congregate care residents and staff. Every hospital will have access to the first vaccine allocation.
The CEO of New York-based Pfizer, Albert Bourla, has promoted a very high 90.1% effective percentage of their vaccine. According to the FDA, the most commonly reported side effects, which typically lasted several days, were pain at the injection site, tiredness, headache, muscle pain, chills, joint pain, and fever. Both companies, Pfizer and BioNtech, are expected to produce globally up to 50 million vaccine doses in 2020 and up to 1.3 billion doses by the end of 2021.